Today in class we began to look at the first 4 of the 5 types of reactions. These 4 types are synthesis, decomposition, combustion, and single replacement.
Synthesis sounds like the name this is where we take 2 small compounds or 2 elements and add them together to make one larger compound.
Decomposition is the exact opposite of the a synthesis reaction and is where we take a larger compound and break it down into its smaller parts.
Combustion is a reaction between a hydrogen and carbon (sometimes oxygen) containing compound and oxygen to always produce carbon dioxide and water.
Single Replacement is a reaction where a metal/halogen will replace a metal/halogen in a compound if they are more reactive.
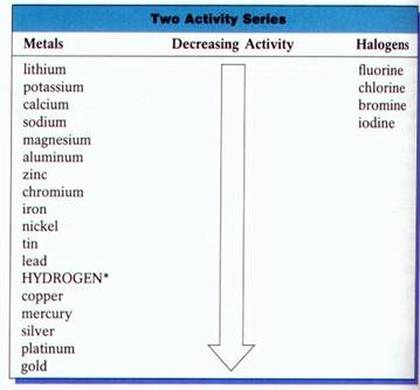
To determine if a single replacement reaction will occur by using a activity series. If the metal being added to the compound is higher on the list than the metal in the compound then the metal will replace it.
Synthesis sounds like the name this is where we take 2 small compounds or 2 elements and add them together to make one larger compound.
Decomposition is the exact opposite of the a synthesis reaction and is where we take a larger compound and break it down into its smaller parts.
Combustion is a reaction between a hydrogen and carbon (sometimes oxygen) containing compound and oxygen to always produce carbon dioxide and water.
Single Replacement is a reaction where a metal/halogen will replace a metal/halogen in a compound if they are more reactive.
To determine if a single replacement reaction will occur by using a activity series. If the metal being added to the compound is higher on the list than the metal in the compound then the metal will replace it.
|
| ||||
 RSS Feed
RSS Feed
